CARS 2015: International Conference and Exhibition on Computer Assisted Radiology and Surgery

The 29th International Conference and Exhibition of Computer Assisted Radiology and Surgery is taking place in Barcelona, Spain. This conference is an annual event that brings together an international community of scientists, engineers and physicians to present and discuss research and development work on novel algorithms, systems and applications focused on radiology and surgery. Kitware has been an active participant of this conference for many years and this year is no different. Kitware engineers’ Ricardo Ortiz and Dzenan Zukic are participating in the conference and they are scheduled to present three papers.
Paper 1:
SlicerPET: A workflow based software module for PET/CT guided needle biopsy: Dženan Zukić, Julien Finet, Emmanuel Wilson, Filip Banovac, Giuseppe Esposito, Kevin Cleary, and Andinet Enquobahrie
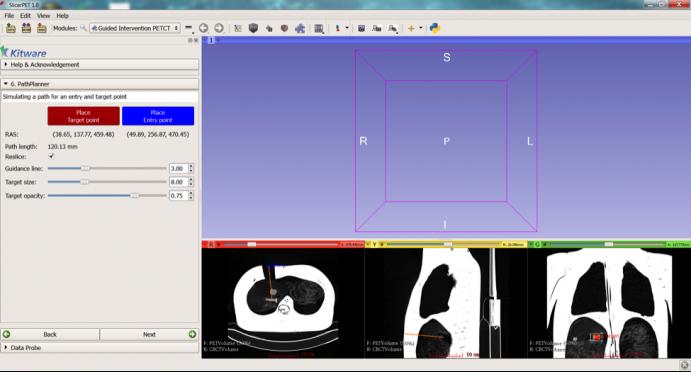
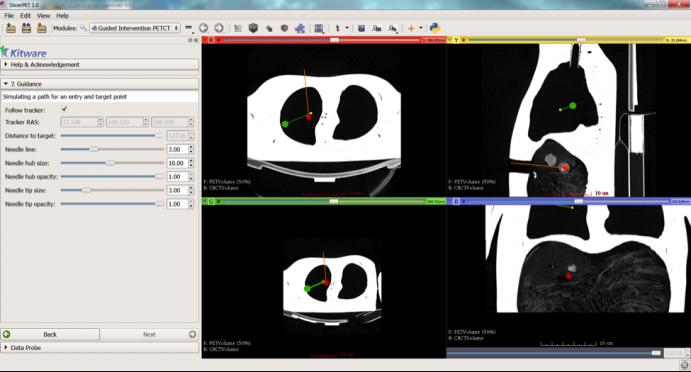
Dženan Zukić will present this paper focsed on SlicerPET: a user-friendly workflow based module developed using open source software libraries and the 3D Slicer platform to guide needle biopsy in the interventional suite. Biopsy is commonly used to confirm cancer diagnosis when radiologically indicated. Given the ability of PET to localize malignancies in heterogeneous tumors and tumors that do not have a CT correlate, PET/CT guided biopsy may improve the diagnostic yield of biopsies. To facilitate PET/CT guided needle biopsy, we developed a workflow that allows us to bring PET image guidance into the interventional CT suite. The figure below shows two steps in the workflow: Path planning on fused PET-CT image (Top) and Needle-guidance step (bottom).
This work was supported by the National Cancer Institute of the National Institutes of Health under Award Number R42CA153488. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Paper 2:
Postoperative Evaluation of Craniosynostosis based on 3D Statistical Shape Model: Jin Qi, Dženan Zukić, Benjamin Wood, Gary Rogers, Christopher Meyer, Ricardo Ortiz, Carlos S. Mendoza, Andinet Enquobahrie, and Marius George Linguraru
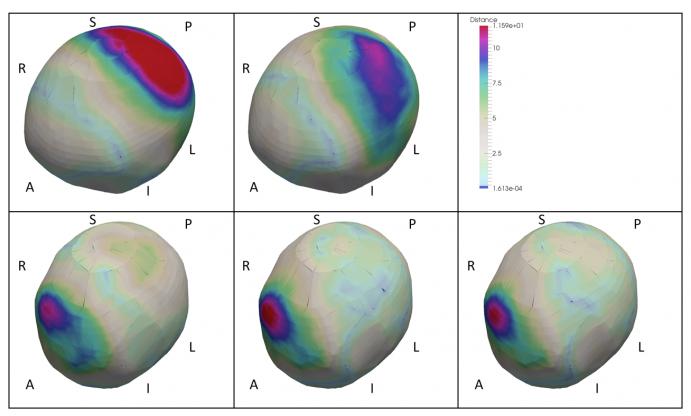
This is a collaborative work with Children National Medical Center. Dzenan will present methods developed to enable the precise, quantitative comparison of cranial shape before and after cranial vault reconstruction to determine the efficacy of specific reconstructive techniques. Craniosynostosis is caused by premature fusion of cranial sutures resulting in cranial malformation. Surgical treatment planning of craniosynostosis is mostly qualitative, subjective and irreproducible guided mainly by the surgeon’s experience. This work focuses on development of a virtual surgical planning system to enable quantitative evaluation and precise surgery. Objective postoperative evaluation of cranial shape could provide important information on the success of the surgical procedure. Point-wise distance were computed between the identified closest normal shape (CN), normal mean shape (MN), pre-operative (Pre) and actual post-operative CT scan (Post). After surgery, mean deformations over the cranium were improved by 24.12% in relation to the closest normal shape and 39.87% in relation to the mean normal shape. These improvements were particularly striking on the left parietal bone, 79.01% vs. 74.33% in relation to the closest and mean normal shapes, respectively. These results are in agreement with the clinical observation that the left parietal is one of the most affected by left coronal synostosis. The figure below shows representative visualizations (camera position is above, in front and to the left of the head). Top left: Pre-MS. Bottom left: Post-MS. Top center: Pre-CN. Bottom center: Post-CN. Bottom right: Post-Pre. A-P, L-R and S-I: anatomical orientation. Distance is in millimeters.
Paper 3:
Stress analysis of cranial bones for craniosynostosis surgical correction: Ricardo Ortiz, Dženan Zukić, Jin Qi, Benjamin Wood, Gary F. Rogers, Andinet Enquobahrie, and Marius George
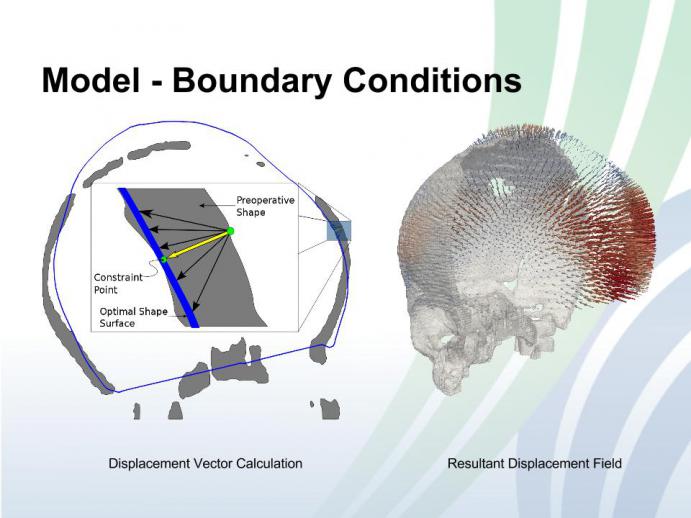
In current practice, the decision of performing surgical correction for patients with craniosynostosis is typically based on subjective visual assessment of the severity of the cranial malformation. These decisions have a major impact on the management of these young patients requiring surgical intervention to correct malformation in the cranial bones and reduce intracranial pressure. The bone correction procedure can be simulated via biomechanical modeling based on the physical properties of the bone. The resulting desirable post-correction shape and the stress of the bone can then be used to evaluate and compare individual bone correction plans before carrying out the actual surgeries, as well to quantitatively analyze the efficacy of specific reconstruction techniques. Depending on the bone rigidity, which is highly correlated to children’s age, the cranial bone might fracture due to the increased amount of pressure during the surgery. Therefore, stress measurements are important to evaluate surgical plans and avoid undesirable bone fracture incidents. Currently there are no available surgical planning systems to optimize intervention and assist in the reconstruction of the cranial vault. Our aim is to provide objective biomechanical modeling tools to facilitate evaluating and optimizing surgical plans for craniosynostosis correction. Figure below shows a two dimensional profiles of the preoperative shape and the optimal shape surface. In the zoomed-in-in region, a displacement vector (yellow) is obtained by averaging a sub-sampled set of displacement vectors (black). This vector is then used to obtain constraint points on the optimal model surface. The two points (in green) are used to create a boundary condition on the surface of the preoperative shape. Right figure shows a mesh generated for the preoperative shape and a sub-sampling of the computed displacement vectors using the optimal shape
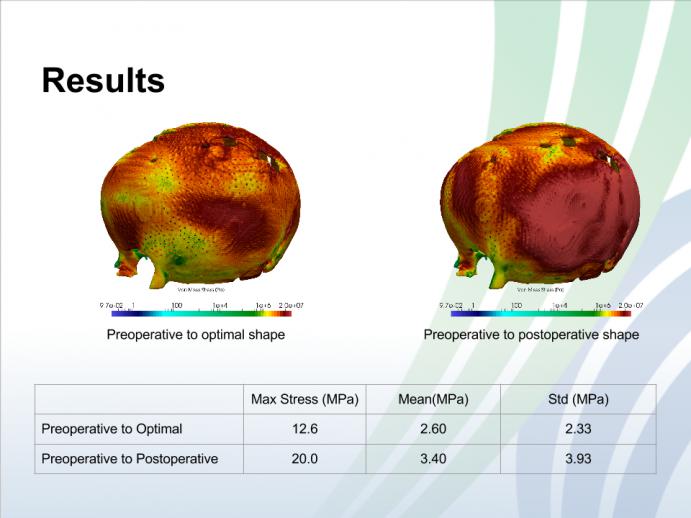
Figure below shows stresses computed for displacements generated between the preoperative model and the optimal shape are smaller than the stresses computed between the preoperative and postoperative models.
This work was supported by the Eunice Kennedy Shriver National Institute Of Child Health & Human Development of the National Institutes of Health under Award Number R41HD081712. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
If you would like to set up a time to meet with Ricardo and Dzenan at the conference to discuss employment opportunities, potential collaboration or consulting services, please contact our corporate office by phone (518) 371-3971, or email at kitware@kitware.com.