Traumatic Brain Injury Assessment: Automating Optic Nerve Sheath Diameter Estimation

One driving application of our point-of-care, computer-assisted ultrasound system is in-field traumatic brain injury (TBI) assessment via the detection of increased intracranial pressure. Delayed treatment of increased intracranial pressure can cause temporary or permanent brain damage or even long-term coma and death. For example, it has been shown that acute subdural hematomas in severe TBI patients cause significant increase in intracranial pressure, are associated with 90% mortality if detected and treated more than 4 hours after injury, and yet are associated with only 30% mortality if detected and treated earlier.
Our system detects elevated intracranial pressure associated with TBI by automatically measuring the diameter of the optic nerve sheath, a known indicator of elevated intracranial pressure ( see for example this Emcrit blogpost ).
Algorithm
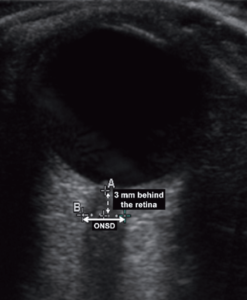
In a clinical setting the optic nerve sheath diameter is measured by ocular ultrasound (see Image). An ultrasound image is acquired and the physicians manually measures the diameter of optic nerve sheath at a location 3mm behind the retina. Acquiring such images and making these measurements is challenging and time consuming.

Our algorithm reports estimates of the optic nerve sheath diameter in near real time as an ultrasound probe is swept across the closed eye of a patient. At a high level, the algorithm proceeds by locating the orb of the eye through registration of an ellipse with the largest dark circle in the image data. From the ellipse an approximate location of the optic nerve is constructed and used to fit two bars to the walls of the optic nerve. This high-level description leaves out several intermediate steps such as those involving image smoothing, morphological operations, and distance transforms, that are required to achieve good registration results. The algorithm runs on a variety of ultrasound images from different probes and with large variations in image quality.
This algorithm is now integrated into a user-friendly interface for use with Interson ultrasound probes on any windows platform. The algorithm performs estimates at 4 frames per second. Depending on the ultrasound probe, the algorithm can be simplified by skipping the eye estimation step to run at around 20 frames per second.

Code and binary releases of the algorithm and graphical user interface are available on github:
- Platform-independent command-line executable to run on single images [github]
- Interactive graphical user interface for use with Interson probes [github]
Evaluation
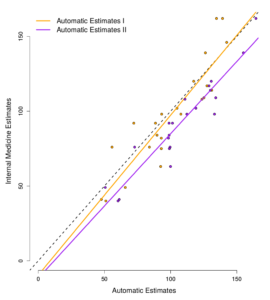
To evaluate the quality of the estimates we had 13 volunteers ranging from Kitware engineers to medical professionals annotate 23 ultrasound images. The image below shows a linear fit of the automatic estimated diameter from two different parameter settings of the algorithm as compared to a medical professional. The automatic estimates had an R2 of around 0.9 and 0.8, respectively. The mean intra correlations among medical professionals was 0.84. Thus, on this small sample size the automatic estimates perform on par with medical experts. The correlation among experts matches results of previous studies [2,3].
For additional evaluation of the performance of the algorithm we built a phantom from gelatine in which 3D-printed “optic nerves” (plastic discs) of known diameter can be embedded under gelatine orbs and imaged, producing ultrasound images that look surprising like clinical ocular ultrasound images.

The goal of this evaluation is to check if an accurate estimate of the optic nerve is possible with the proposed algorithm. In this controlled setting we first located the optic nerve using the ultrasound. Once a good image of the optic nerve was achieved the automatic estimation process was started and run interactively for about 10 seconds which resulted in approximately 40 to 50 optic nerve sheath diameter estimates. The table below shows that the means of the automatic estimates are within less than +/- 0.5mm with a relatively tight distribution of estimates around the ground truth diameter. A study on intra-operator variations on the same type of phantom, reports standard deviations of the measurements ranging from 0.25 to 0.6 mm depending on diameter size.
| Disc Size | Mean of Estimated Size | Range of Estimated Size (Quartiles) |
| 7mm | 7.0 | 6.4 – 7.7 |
| 6mm | 6.1 | 5.8 – 6.6 |
| 5mm | 5.1 | 4.4 – 5.7 |
| 4mm | 4.4 | 4.2 – 4.5 |
| 3mm | 3.4 | 2.9 – 3.5 |
Results of automatic estimates on gel phantom
Conclusion
The results presented indicate that the algorithm performs well in a controlled setting. A retrospective analysis of clinical images indicate that our system performs similar to an expert, and a phantom study using 3D printed optic nerves of known diameter suggests that our system is accurate.
The next step is to integrate an image quality estimation algorithm, so that high quality images of the optic nerve can be automatically identified as a probe is swept over the eye. This will eliminate the need for the operator to view, interpret, or make measurements on an ultrasound image when using it to assess a dilated optic nerve sheath, as indicative of increased intracranial pressure. Ultimately, we aim for a system that can be used by novice operators with minimal ultrasound experience. The system will report when a sufficient image has been acquired, and it will automatically determine if the diameter of the optic nerve is greater than 5mm, thereby indicating increased intracranial pressure associated with mild or more severe TBI [4].
The long term goal of Kitware is to develop a lightweight portable ultrasound system that, in addition to automated diagnosis of TBI, includes automatic diagnosis tools for pneumothorax (detached lung) and internal bleeding.
Acknowledgements
This work was performed in collaboration with Sean Montgomery, M.D. (Surgery, Duke University Medical Center), Bradley Freeman, M.D. (Surgery, Washington University School of Medicine), and Deborah Shipley Kane, M.D. (Emergency Medicine, Washington University School of Medicine).
This work was funded, in part, by the following grants:
- NIH/NIBIB: “In-field FAST procedure support and automation” (R43EB016621)
- NIH/NINDS: “Multimodality image-based assessment system for traumatic brain injury” (R44NS081792)
- NIH/NIGMS/NIBIB: “Slicer+PLUS: Collaborative, open-source software for ultrasound analysis” (R01EB021396)
References
[1] Dubost, Clément, et al. “Optic Nerve Sheath Diameter Used as Ultrasonographic Assessment of the Incidence of Raised Intracranial Pressure in PreeclampsiaA Pilot Study.” The Journal of the American Society of Anesthesiologists 116.5 (2012): 1066-1071.
[2] Johnson, Garrett GRJ, et al. “Estimating the accuracy of optic nerve sheath diameter measurement using a pocket-sized, handheld ultrasound on a simulation model.” Critical Ultrasound Journal 8.1 (2016): 18.
[3] Zeiler, F. A., et al. “A unique model for ONSD Part II: inter/intra-operator variability.” Can J Neurol Sci 41 (2014): 430-435.
[4] Maissan IM , Dirven PJ , Haitsma IK , Hoeks SE , Gommers D , Stolker RJ “Ultrasonographic measured optic nerve sheath diameter as an accurate and quick monitor for changes in intracranial pressure.” J Neurosurg. 2015 Sep;123(3):7437