MHSRS facilitates collaboration between military providers, academia, and industry on medical and health care research. The topics that will be covered at the symposium include combat casualty care, military operational medicine, clinical and rehabilitative medicine, medical simulation and information sciences, military infectious diseases, and the radiation health effects.
Kitware is excited to be exhibiting at MHSRS 2023 to showcase our innovative medical and biomedical software. In our booth, we will be giving demos of our medical modeling and simulation, point-of-care ultrasound (POCUS), craniomaxillofacial surgical, and medical image segmentation solutions.
To learn more about Kitware’s medical computing expertise and the benefits of using open source technology, send us a message.
Kitware’s Activities and Involvement
In addition to exhibiting, Kitware is proud to have an oral presentation and two posters accepted at MHSRS.
Oral Presentation
Evaluation of Commercially-Available Point-of-Care Ultrasound for Automated Optic Nerve Sheath Measurement
Presenter: Brad Moore, Ph.D.
Measurement of the optic nerve sheath diameter via ultrasonography is a promising, non-invasive metric of intracranial pressure that may be used during in-field patient triage. However, first responders are not typically trained to conduct sonographic exams and/or do not have access to an expensive ultrasound device. We examine the suitability of five commercially-available, low-cost, portable ultrasound devices that can be combined with AI algorithms to reduce the training required for and cost of in-field optic nerve sheath diameter measurement. This work is focused on the quality of the images generated by these low-cost probes. We report results of a clinician preference survey and compare with a lab analysis of three quantitative image quality metrics. We also examine the suitability of the devices in a hypothetical far-forward deployment using operators unskilled in ultrasound, with the assumption of an onboard AI video interpreter.
Poster Session
A machine learning surrogate model for risk assessment associated with electronic nicotine delivery systems
Authors: Matthew Bernstein (Kitware), Rachel Jennings, Ph.D. (Applied Research Associates), Jeff Schroeter, Ph.D. (Applied Research Associates), and Rachel Clipp, Ph.D. (Kitware)
The number of electronic nicotine delivery systems (ENDS), including e-cigarettes and vaping devices, being used by military personnel is increasing significantly, presenting a significant risk to the force health, readiness, and resilience. Most studies focus on the immediate effects of ENDS by measuring the levels of nicotine, carcinogens, and toxicants in the airways, urine, and blood. However, there is a need to better understand and evaluate the risks of long-term use of these products. Since ENDS have only been on the market for approximately fifteen years, the lack of epidemiological data poses challenges to developing predictive risk assessment models. Computational models have also been used to explore the risk of inhalation injury and toxicity, but the expense of many of these models can be limiting. In our poster, we propose using machine learning (ML) algorithms to predict the risk assessment of ENDS constituents using training data produced by our validated computational model, the Pulse Physiology Engine.
Poster Session
ARGUS: An Open-Source Platform for Ultrasound Video AI That Supports Multiple Point-of-Care Applications
Authors: Stephen Aylward, Ph.D. (Kitware), Barry Ravichandran, M.S. (Kitware), Christopher Funk, Ph.D. (Kitware), Brad Moore, Ph.D. (Kitware), Forrest Li, M.S. (Kitware), Jon Crall, Ph.D. (Kitware), Michael J. Morris, M.D. (Brooke Army Medical Center), Jess T. Anderson, Maj, USAF, M.C. (Brooke Army Medical Center), Andrew M. Hersh, M.D. (Montrose); Yuriy Bronshteyn, M.D. (Duke University Hospital), Sean Montgomery, M.D. (Duke University Hospital)
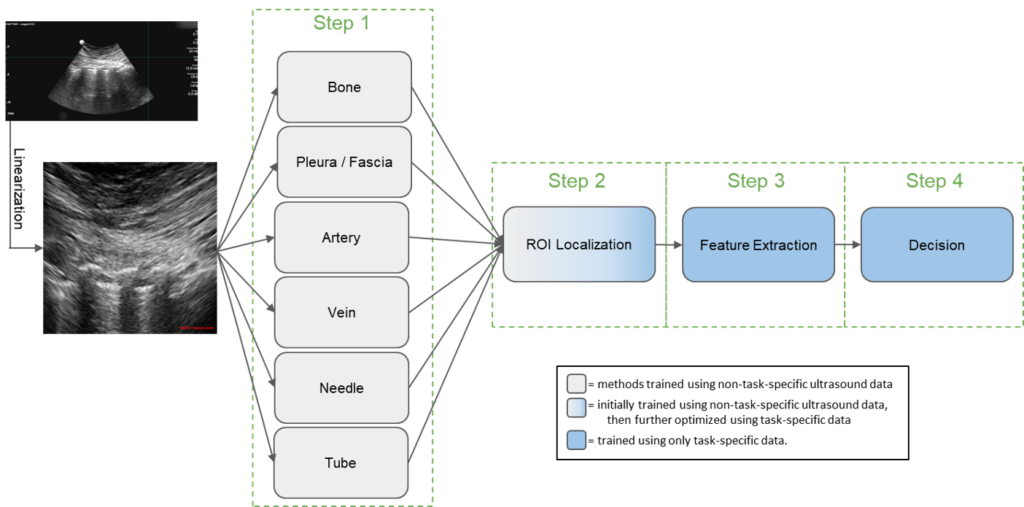
For POCUS to be routinely used in the field, there need to be justifications for its weight, maintenance, and training requirements. One such justification is improving outcomes so the technology can be used in multiple applications. To help POCUS AI achieve multi-application capabilities, we have devised a software platform that implements a POCUS video AI workflow that promotes re-using AI components across multiple applications. Our platform is available as open source software for academic and commercial use. It was developed as part of the DARPA POCUS AI program. In addition to supporting multiple applications, it supports training AI systems using limited data and running AI systems on mobile hardware, with limited computational resources. We have demonstrated its use on four POCUS applications: pneumothorax detection (PTX), peripheral nerve block delivery (PNB), endotracheal intubation confirmation (ETT), and optic nerve sheath diameter estimation (ONSD). Comparisons with ultrasound experts indicate the diverse viability of our platform.
Advancing Science and Technology to Empower Innovation
At Kitware, we believe that accelerating innovation will make the world a safer, healthier, and better-informed place. That’s why we deliver innovative solutions to our customers built on open source technology. Our open source philosophy also offers unique and valuable benefits to our customers and collaborators, saving time and money.
Kitware has experience working with the U.S. government, including the DoD and IC. Our solutions come with unlimited rights to our government customers, including no licensing fees or restrictions. We can provide support at any stage of the medical product lifecycle, from initial R&D to IEC 62304 and HIPAA -compliant product development.
Our development, testing, and documentation practices are aligned with FDA requirements and HIPAA technical safeguards for software products. Kitware can sign NDAs and HIPAA BAAs, and we have the infrastructure to handle projects subject to CUI, EC, ITAR, and HIPAA regulations.
Harness the power of open source technology to efficiently develop cutting-edge medical software.
Physical Event
Gaylord Palms Resort & Convention Center
Kissimmee, FL
